PHARMACY ADVANTAGE PROGRAM
iLet Bionic Pancreas
$0 to Start
First 30 days completely free — no hardware lock-in, no upfront cost.
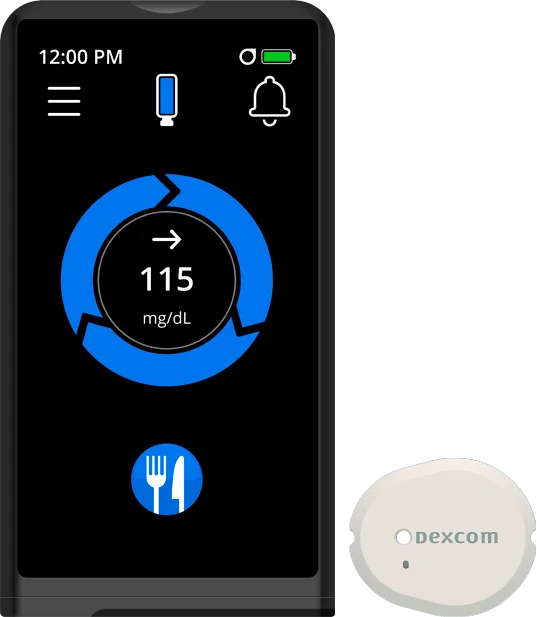
NOW AVAILABLE
The iLet Bionic Pancreas
+ Dexcom G7 15 Day
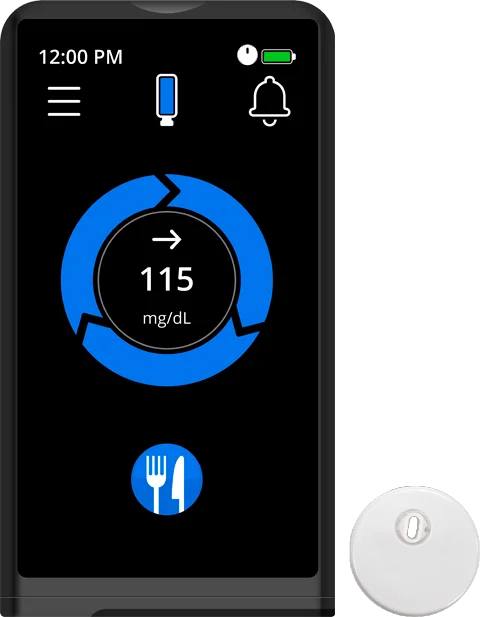
NEW!
iLet + FreeStyle Libre 3 Plus sensor
Now you can use the FreeStyle Libre 3 Plus sensor with your iLet

Introducing
The Bionic Circle
Invite up to 10 friends and family to join your Bionic Circle.




